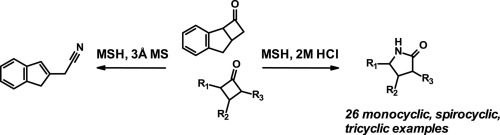
![Multicatalytic Beckmann rearrangement of 2-hydroxylarylketone oxime: Switchable synthesis of benzo[d]oxazoles and N-(2-hydroxylaryl)amides - Tetrahedron Lett. - X-MOL Multicatalytic Beckmann rearrangement of 2-hydroxylarylketone oxime: Switchable synthesis of benzo[d]oxazoles and N-(2-hydroxylaryl)amides - Tetrahedron Lett. - X-MOL](https://xpic.x-mol.com/20180918%2F10.1016_j.tetlet.2018.09.043.jpg)
Multicatalytic Beckmann rearrangement of 2-hydroxylarylketone oxime: Switchable synthesis of benzo[d]oxazoles and N-(2-hydroxylaryl)amides - Tetrahedron Lett. - X-MOL
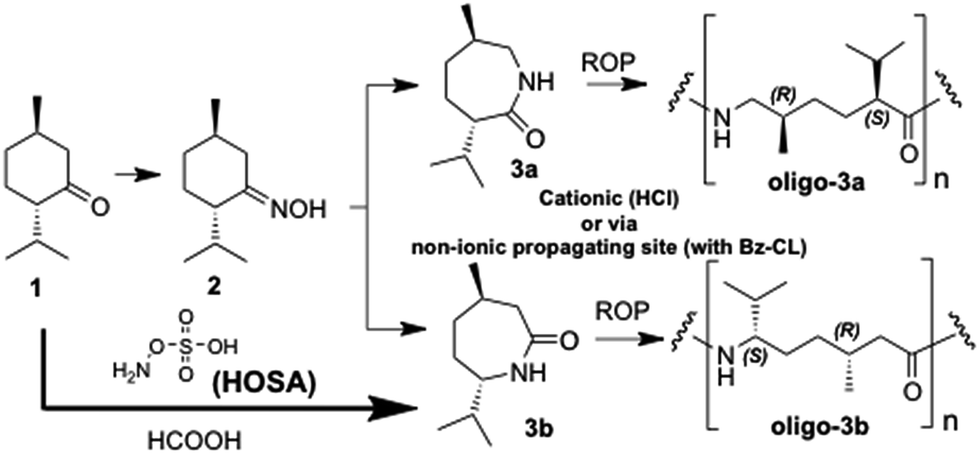
New insights into synthesis and oligomerization of ε-lactams derived from the terpenoid ketone (−)-menthone - RSC Advances (RSC Publishing)
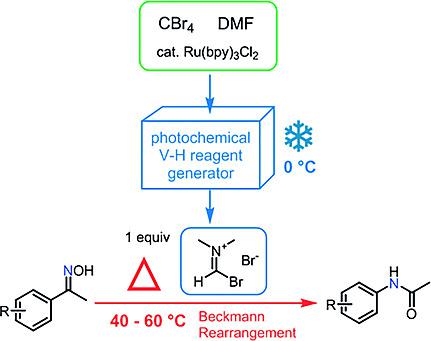
Visible Light‐Promoted Beckmann Rearrangements: Separating Sequential Photochemical and Thermal Phenomena in a Continuous Flow Reactor - Eur. J. Org. Chem. - X-MOL

Figure 1 from Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). | Semantic Scholar
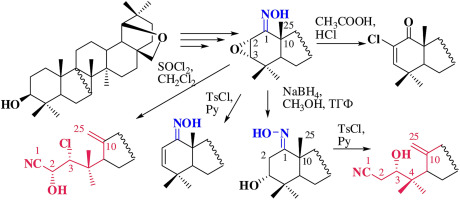
Synthesis of 1,10-seco-triterpenoids by the Beckmann fragmentation from allobetulin,Tetrahedron - X-MOL
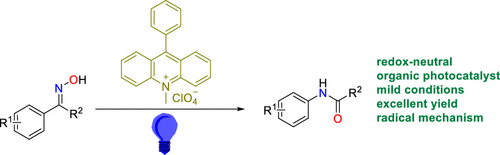
Visible-Light-Induced Beckmann Rearrangement by Organic Photoredox Catalysis.,Organic Letters - X-MOL

Direct and Stereospecific Synthesis of N-H and N-Alkyl Aziridines from Unactivated Olefins Using Hydroxylamine-O-Sulfonic Acids. - Abstract - Europe PMC

Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). - Abstract - Europe PMC

Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). - Synthesis - X-MOL

PDF) The HosA Histone Deacetylase Regulates Aflatoxin Biosynthesis Through Direct Regulation of Aflatoxin Cluster Genes

Спрей-пятновыводитель пот и дезодорант Dr.Beckmann, 250 мл, цена 13.20 руб., купить в Минске — Deal.by (ID#111068765)

PDF) The HosA Histone Deacetylase Regulates Aflatoxin Biosynthesis Through Direct Regulation of Aflatoxin Cluster Genes